Achieving Your Gene Therapy Workflow Viability
Published in Cell & Gene, New Economics of Cell and Gene Therapy Parts I and II by Jason C. Foster, Gaurav Shah, and Sanjay Srivastava have focused on a thesis that current methodologies and practices in designing commercial scale processes is not sustainable for the long term success of these promising modalities.
The authors point out today’s product development processes are predominantly manual – resulting in low-efficiency, unclear understanding of the process biology, unpredictable yield, high costs and high failure rates; and that with each new cell or gene therapy that is priced at $400K-$4M per patient, we are limiting our ability to get widespread coverage and early line usage for these therapies with these products frequently struggling to achieve scale.
The authors conclude that the CGT industry cannot continue to develop and launch products that are safe and effective but not commercially viable, or we will risk the future of this promising modality.
What elements make up viability?
Reasonable COGS to achieve target profit margins at an acceptable price point
TPP that provides the likelihood of target line coverage (i.e. market access)
Manufacturing process that can be scaled to meet the anticipated peak demand
Supply expansion strategy for earlier line coverage/indication expansion/global access
But how does a company partner or internally develop viability of COGs, TPP, and Manufacturing process scalability in their workflow design? N+1’s Process Engineering Team believes that we can help our clients achieve this level of viability by bringing back an often overlooked habit of Highly Successful People or in this case Organizations:
“BEGIN WITH THE END IN MIND”
Our Process Engineering and Workflow Team is focused on helping our clients start their process design conversation at a repeatable, scalable, quality focused workflow for all advanced therapeutic modalities leveraging years of experience as Process Engineers at commercial scale where we have redesigned or supported manufacturing colleagues as they were forced to live with manual, highly variable, and ultimately poorly scalable advanced therapeutic processes.
Thinking ahead, and translating that into tactical, thoughtful, design decisions is a skill that we are committed to helping our clients realize.
In a presentation titled “Death to (Tubing) Welding” for ISPE Boston I presented a case study in translating organizational capacity goals to commercially viable process engineering design and process changes and its impact to COGs for high throughput gene therapy manufacturing processes.
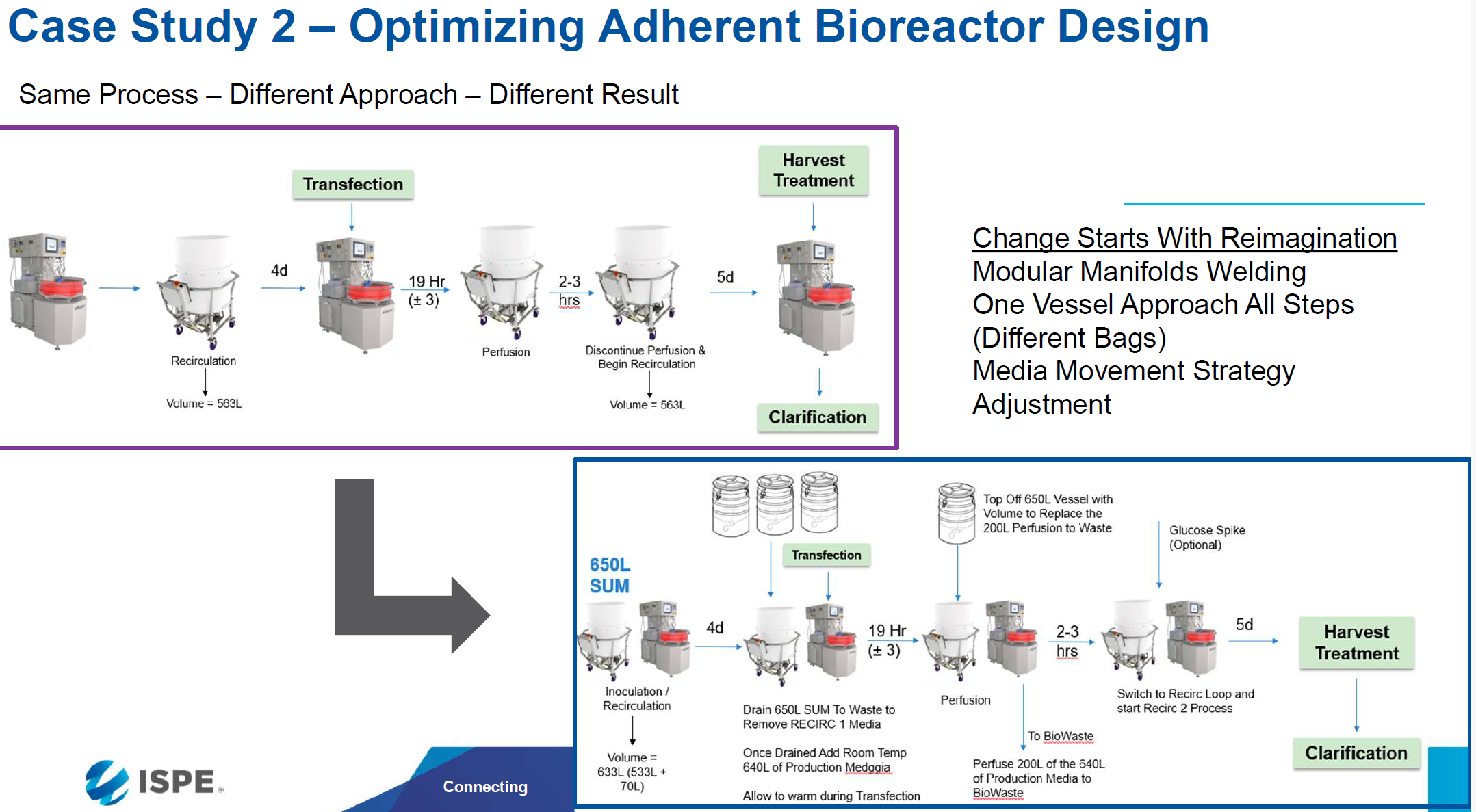
In this case study, due to process variability in the transfection step the client was pooling multiple upstream batches prior to progressing the process through purification. By beginning with the end in mind and looking at the proposed commercial scale workflow we were able to work with the client to update the proposed lot pooling strategy to support a risk reduced commercial manufacturing process as well as streamline their proposed adherent bioreactor process to reduce the cost of COGs, setup time, and manufacturing footprint for their high throughput commercial scale gene therapy workflow Figures 1-4.
Figure 1: Capacity Modeling and Pooling Strategy Adjustment Analysis
Figure 2: Initially Proposed Process From Client Development
Figure 3: Optimized Adherent Bioreactor Design for Gene Therapy Client
Figure 4: Final Commercial Scale Adherent Process
This Client case study highlights that it is possible to partner with organizations equally committed to CGT commercial scale viability and achieve drastic results with targeted, thoughtful shifts in process and workflow engineering design.
For this client optimizing their adherent reactor design resulted in a new process that saved over 374K in capital equipment costs to implement, reduced the manufacturing footprint of each instance of the unit operation by 104 sq ft and reduced the manufacturing setup and execution time of this process from 3-4hrs to 20-30 minutes.
The case study from N+1’s Process Engineers proves that Jason C. Foster, Gaurav Shah, and Sanjay Srivastava’s call to enhanced commercial viability for cell and gene therapy workflows:
IS ACHIEVABLE FOR ALL ORGANIZATIONS
What can we partner and do together?
Full Link to Cell and Gene Article Referenced: https://www.cellandgene.com/doc/new-economics-of-cell-and-gene-therapy-part-ii-0001?trk=feed_main-feed-card_feed-article-content